Called Author Invited Author
DOI: 10.4274/Turk Ped Ars.45.23
Inguino-scrotal pathologies / Inguino-scrotalpathologies
Ünal Zorludemir
Çukurova University Faculty of Medicine, Department of Pediatric Surgery, Department of Pediatric Urology, Adana, Turkey
Abstract
Inguinal and scrotal pathologies are the most common surgical problems in childhood. Most of these are inguinal hernia, hydrocele, undescended testis and testicular tumors. The most common pathologies causing acute scrotal pain and/or swelling are testicular torsion, epididymo-orchitis and appendix testicular torsion. Timely diagnosis and treatment of these disorders is of vital importance. (Turkish Ped Arş 2010; 45 Special Issue: 23-8) Journal of the Turkish Archives of Pediatrics,
Key words: Acute scrotum, child, hydrocele, inguinal hernia, undescended testis, testicular tumor
Summary
Inguinal and scrotal pathologies are the most common surgical problems in childhood. Most of these pathologies are inguinal hernia, hydrocele, undescended testicle and testicular tumors. Among the most common pathologies that cause acute scrotal pain and/or swelling are testis torsion, epididymo-orchitis and torsion of appendix testis. The timely and correct diagnosis and treatment of these disorders in time is of vital importance. (Turk Arch Ped 2010; 45 Suppl: 23-8)
Key words: Acute scrotum, child, hydrocele, inguinal hernia, testicular tumor, undescended testicle
Inguinal hernia (inguinal hernia)
It is the most common indirect inguinal hernia in the groin in children. It develops when the peritoneal extension, called the processus vaginalis, remains open.
It is seen in 0.8-4.4% of term babies (1) and between 16-25% in premature babies (2,3). Although the opening of the processus vaginalis is congenital, the clinical age of hernia varies according to the width of this opening. Since the processus vaginalis closes later, it is more common on the right (60%), and is bilateral at a rate of 10% (4).
The typical history is swelling that occurs from time to time in the groin or in the scrotum in boys and in the labium majus region in girls (Picture 1,2). This swelling becomes evident in situations that increase intra-abdominal pressure such as crying and straining, and disappears by lying down or pushing.

Figure 1. Inguinal hernia in a boy

Figure 2. Inguinal hernia in a girl
For the examination of inguinal hernia, the environment should be warm, both sides should be observed at the same time to examine whether there is swelling and to evaluate whether the testicles are in the scrotum. For older children, straining as if they are pooping can help to see the swelling. If there is a plug, it should be tried to be pushed towards the inguinal region, if it cannot be pushed, it should be evaluated whether it is mobile (lymph gland, cord cyst, testis) or fixed (strangulated hernia). Despite all these, swelling due to hernia may not be seen during the examination. In such cases, the family should be questioned where they saw the swelling and should be told to show the location of the swelling.
Inguinal hernia does not go away on its own, there is always a risk of suffocation. This may cause compression of the intestine and/or testis in boys and more often ovary and/or tuba uterine in girls, necrosis and gangrene development in these organs, and even sepsis, resulting in death of the patient. The younger the child and the hernia, the greater the risk of choking. For these reasons, the treatment of an inguinal hernia is ‘surgery as soon as possible under elective conditions when the diagnosis is made’. What is meant by elective conditions here is the child’s absence of an infection, anemia, or other systemic disorder that would prevent surgery and/or general anesthesia.
Hernia strangulation risk is higher in premature babies (31%), and it is recommended to repair the inguinal hernia before being sent home from the neonatal intensive care unit (5).
In a child with an inguinal hernia on one side, the probability of having a hernia on the opposite side in the future is 20% (6-8). Routine surgery of the contralateral side without any sign of hernia is not as common as it used to be.
Hernia strangulation (incarceration) is the inability of the organs to be stuck in the hernia sac and pushed into the abdomen. This event may progress to necrosis and gangrene of the entangled intestines, testis in boys, and ovary and/or tuba uterine in girls, and perforation of the intestine. A strangulated hernia is confused with hydrocele and sometimes with testicular torsion. The absence of pain in the history and the persistence of swelling for a long time should primarily suggest hydrocele. Inguinal or scrotal swelling that occurs suddenly when there was no swelling before should first suggest a strangulated hernia. In the differential diagnosis of scrotal swelling in men, the most practical diagnostic method is standing direct abdominal radiography, which also includes the scrotum (Picture 3). The presence of gas-liquid level in the scrotum indicates that there is an intestine here, thus a hernia. Ultrasonography (USG) is performed if necessary for differential diagnosis.

Figure 3. Scrotal hernia, standing direct abdominal X-ray
Findings suggestive of malnutrition in the compressed organ in a patient with strangulated hernia (edema, color change, local and/or If there is systemic fever, leukocytosis), emergency surgery is required.
Congenital hydrocele (9)
Hydrocele is a collection of fluid between the inner and outer tunica vaginalis layers surrounding the testis and usually occurs at birth. It can be associated with the peritoneal cavity (communicating) or unrelated (non-communicating, simple). When communicating, swelling decreases when the baby is lying down, but does not disappear completely, and increases when standing up. It is more common on the right, often bilateral. It is found in many children with an inguinal hernia.
In case of segmental closure of the processus vaginalis, localized cord hydrocele (cord cyst, Nuck’s canal cyst in girls) develops. This may be associated or unrelated to the peritoneal cavity. It is a painless swelling with cord structures. It may be just above the testis or in the inguinal region. The mass is mobile and transmits light. USG can be helpful in diagnosis (10,11).
Hernia and hydrocele can usually be distinguished from each other by history and examination. Hernia swelling occurs with increased intra-abdominal pressure and from time to time. It disappears completely by itself or by suppression. Hydrocele swelling is mostly present since birth and does not disappear completely in a short time, it does not go in with pressure. However, if the hydrocele is associated with the peritoneum (communication), the tension of this swelling decreases when the child is lying down, and becomes more pronounced when awake and standing. Transillumination (light) examination is useful for differentiating solid masses such as testicular tumors from hydrocele and hernia. The most useful method for differential diagnosis in girls is USG.
In most of the congenital hydroceles unrelated to the peritoneum, the hydrocele fluid is absorbed by the age of 1-2 years and disappears. If it does not go away, surgery is performed. Apart from that, hydroceles with hernia and abdominoscrotal giant hydroceles are operated without waiting. Aspiration of hydrocele fluid is contraindicated in infants, there is a risk of infection (11).
Undescended testis
The testicles extend into the scrotum from the 23rd week of pregnancy following the peritoneal extension called the processus vaginalis. It is one of the most common anomalies at birth and is found in 3% of term newborn boys (12-15). This rate is 30.3% in premature infants (12). Low birth weight, black or Hispanic race, family history of undescended testicles and cola intake during pregnancy are other factors that affect late testicular descent (16-17).
Undescended testis is when the testis stays on the descent path between the inferior pole of the kidney and the scrotum, where it first develops. Cryptorchidism (hidden testis) is a broader term. The absence of the testis in the scrotum is cryptorchidism. Besides the undescended testis, cryptorchidism also includes ectopic testis and anorchids (absence of testicles). If the testis is in a place other than the descending path, it is called ectopic testis, it can be in the inguinal region, opposite scrotum, perineum, thigh, prepenile region.
An accurate molecular and genetic process to explain the cause of an undescended testis is not yet known.
Approximately 20% of undescended testicles are not palpable. If the testis is not palpable, the possibilities are: small (atrophic), intra-abdominal, deep in the inguinal canal, or no testis on that side (vanishing testis).
Complications of undescended testis can be counted as infertility, malignancy, easier exposure to trauma, torsion and negative psychological impact. Especially for bilateral undescended testicles, the infertility rate is high. Early descent of the testis not only reduces the risk of malignancy, but also enables easier monitoring in the scrotum (18).
Undescended testis has no chance of descending into the scrotum after one year of age. For this reason, in a child over one year old, the testis should be surgically lowered into the scrotum without waiting.
There are also groups that reduce the download process to six months. Removing the undescended testis (orchiectomy) after adolescence is the most widely adopted method.
Retractile testis (shy testis) is the fact that the testis spontaneously escapes from the scrotum into the inguinal canal due to the active cremaster reflex, but can be easily lowered into the scrotum by hand and remains in the scrotum when released. It is most common between the ages of 3-7. It is a physiological event. If the testis can be easily lowered to the lower end of the scrotum, no treatment is required (19). Despite being lowered, there is a 50% risk that the testis will later go up in children whose testicles cannot easily stay in the scrotum and are tense. These children require annual follow-up until the testis is completely scrotal.
Ascending testis is a condition in which the testis escapes into the inguinal region, although it was previously seen in the scrotum, that is, descended. Due to the decrease in the number of germ cells, it is recommended to evaluate them as undescended testicles (20).
Testicular tumors (21)
Testicular tumors constitute 1-2% of all childhood solid tumors and are seen at most 2 years of age (22,23). Benign lesions constitute the majority (74%) of testicular tumors in children (24).
The incidence of gonadal tumors increases in patients with intersex conditions (25). There is a risk of tumor formation in 25% of children with mixed gonadal dysgenesis (26), and the incidence increases with age (27). All ‘streak’ gonads in patients with gonadal dysgenesis should be removed (28).
In children with undescended testicles, neoplasia most often develops in bilateral undescended testicles. Inguinal or abdominal undescended testicles left in place have a very high risk of seminoma (74%); There is no risk of testicular cancer in the opposite normal descended testis (29).
The most common finding in children with testicular tumor is ‘painless scrotal solid mass’ (30-32). A painless solid mass detected in the scrotum should be considered malignant until proven otherwise and should be examined (32,33).
USG is routinely preferred in the evaluation of children with testicular tumors (34). There are no USG features that can reliably distinguish between benign and malignant tumors. Findings of anechoic cystic lesions may suggest benign lesions.
It is important to measure AFP in serum in addition to USG. Yolk sac tumors always produce AFP and all AFP positive tumors are thought to contain Yolk sac element (35). AFP level is normal in benign lesions.
Teratoma is the most common testicular tumor in prepubertal children (48%), followed by epidermoid cysts (24,36,37). Prepubertal mature teratomas have a benign clinical course compared to adults. Testicular teratomas are removed with testicular-sparing surgical methods (36-42).
Leukemia and lymphomas are the most common malignancies that spread to the testicles in children. Testicular involvement is seen in 4% of children with Burkitt’s lymphoma and may be the first clinical finding (43).
Varicocele (11)
Spermatic cord veins (pampiniform plexus) are wide and curved. It occurs in approximately 15% of adolescent men. 90% of varicoceles are on the left side (44). Varicocele development is attributed to increased venous pressure in the left renal vein, collateral venous anastomoses, and insufficiency of the valves of the internal spermatic veins. As a result of the toxic effect of varicocele, histological disorders can be seen in both testicles. Since semen analysis is rarely performed in adolescents, testicular volume is evaluated when planning treatment.
Adolescent varicocele is mostly asymptomatic. Most are noticed during routine examination. Such a painless scrotal mass should be differentiated from inguinal hernia, communicating hydrocele, omental hernia, cord hydrocele, epididymal cyst (spermatocele), and scrotal hydrocele. The physical examination should be performed in a warm environment, and the examination should be repeated both lying down and standing, with or without the Valsalva maneuver. It is located on or around the testicle. Its classic appearance is through the skin of the scrotum, like a pack of worms (grade III). In the examination, it is important to evaluate the size of the testis with an orchidometer or an USG examination (11).
A volume loss of more than 2 ml or more than 20% on the side with varicocele is considered sufficient criteria for the surgical treatment of varicocele (45).
Acute scrotum (11)
Pain, tenderness, or swelling that occurs acutely in a child or adolescent is an emergency and should be promptly evaluated for differential diagnosis. Because adolescents do not always understand the potential significance of acute scrotal swelling, their subacute or even chronic scrotal swelling may require urgent evaluation.
Pathology list that can be counted for the differential diagnosis of acute scrotum: spermatic cord torsion, appendix testicular torsion, appendix epididymis torsion, epididymitis, epididymo-orchitis, inguinal hernia, communicating hydrocele, hydrocele, trauma/cord insecticide bites, dermatological lesions, inflammatory vasculitis (Henoch-Schönlein purpura), idiopathic scrotal edema, tumor, spermatocele and varicocele, non-urogenital pathologies (such as adductor tendinitis). It is very important to distinguish spermatic cord torsion, which requires emergency surgery, from others.
Spermatic Cord Torsion (Intravaginal) (11)
It is a true surgical emergency. The reason is the lack of fixation of the testis and epididymis to the fascia and muscle structures in the scrotum (Picture 4). Its classic appearance is sudden onset scrotal pain. In some children this pain is gradual, in others it is very mild. Most children with acute scrotal pain have a previous history of severe, self-limiting scrotal pain and swelling. This is probably due to the previous transient torsion. It may be accompanied by nausea and vomiting. There may be pain in the lower quadrant of the abdomen on the same side. Dysuria and other bladder symptoms are usually absent.
In the differential diagnosis of acute scrotum, the physical examination finding is much more striking than the history. As a result of torsion, that side may be above the testicle scrotum. The cremaster reflex is almost never absent (46).
Color Doppler USG helps by showing the absence of testicular blood flow. Testicular scintigraphy is less reliable. Once diagnosed, treatment is very urgent surgery. If the operation is delayed after six hours, the testis cannot be saved most of the time.
In patients with a history suggesting temporary torsion of the spermatic cord, surgical treatment is recommended for both testicles in elective conditions (47).

Figure 4. Spermatic cord torsion
Torsion of testis and epididymal appendages (11)
Appendix testis müllerian duct remnant, appendix epi- didymis is the wolffian duct residue (Picture-6). Both are prone to torsion as a result of hormonal stimuli during adolescence. The torsion of these structures can vary from insidious onset of scrotal malaise to a clinical picture similar to the acute picture seen in cord torsion. The cremaster reflex does not disappear, the testis is mobile. Where the torsion is, there is the ‘blue dot mark’. Doppler USG and testicular scintigraphy show normal or increased blood flow. If the diagnosis of appendiceal torsion is definite, there is no need for surgical treatment. It is left to self-correct. If indistinguishable from cord torsion, exploration is required.
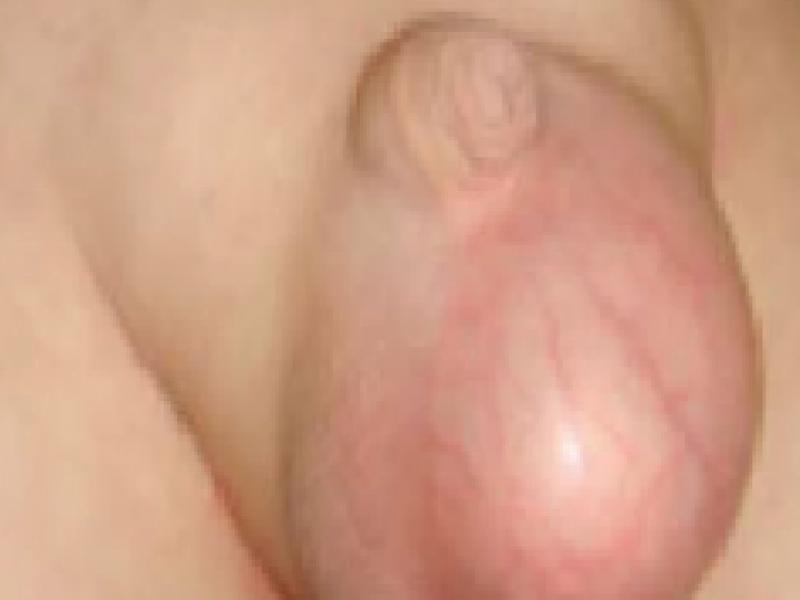
Epididymitis, epididymo-orchitis, orchitis (11.48)
Epididymis and testicular inflammation or infection are as common as acute testicular torsion (48,49). Clinical findings are scrotal swelling, redness and pain (Picture 5). Appendiceal torsion can be confused with epididymitis. Findings in epididymitis can range from swelling and tenderness localized to the epididymis to inflammation involving the entire half of the scrotum. Patients with epididymitis have a cremaster reflex. The absence of this reflex largely supports spermatic cord torsion (44).
Epididymitis is often associated with pyuria, bacteriuria or a positive urine culture. Normal urinalysis does not indicate the absence of epididymitis. Blood flow increased in color Doppler USG and testicular scintigraphy. Mumps etc. Most changes in orchitis or epididymo-orchitis due to viral infections are nonspecific (48,50).
Epididymitis is mostly associated with genitourinary anomalies in young children and infants, and hematogenous spread in older children (48,51). Therefore, in children diagnosed with epididymitis, radiological examinations of the urinary system should be performed to investigate the cause. The younger the child with epididymitis, the more likely it is to have a urinary tract infection, radiological abnormality, or both (52).

Figure 5. Orchitis
Treatment, bed rest, scrotum elevation, local cold or warm application, nonsteroidal anti-inflammatory and analgesic, antibiotics if urinary infection is to be given.
Other causes of acute scrotal swelling
The cause of acute idiopathic scrotal edema is unclear, there is no fever and redness (53). Scrotal tenderness is usually minimal, but pruritus is prominent. Although it is considered to be idiopathic, it can also be caused by allergies, chemical dermatitis, insecticide bites, trauma. Perineal and perianal examination should be performed to distinguish it from perirectal abscess. In most cases, the scrotal wall is thickened and the testicles are palpable. If in doubt, color Doppler USG examination is performed. No treatment is required.
Henoch-Schönlein purpura is systemic vasculitis and may cause scrotal swelling secondary to involvement of the testis, epididymis, or both (54). The cause of vasculitis is unknown. It is seen in 35% of patients. Scrotal findings are usually diffuse, consisting of swelling, redness, and tenderness. Urinalysis may reveal hematuria and proteinuria. Color Doppler USG and testicular scintigraphy show increased blood flow. Although it is usually self-limiting, steroid treatment may be required.
References
1-Brosnther B, Abrams MW, Elboim C. Inguinal hernias in children a study of 1000 cases and review of the literature. Am J Med Womens Assoc 1972; 27:522-5. (Abstract)
2-Rajput A, Gauderer MW, Hack M. Inguinal hernias in very low birth weight infants: incidence and timing of repair. J Pediatr Surg 1992; 27:1322-4. (Abstract)
3-Walsh SZ. The incidence of external hernias in premature infants. Acta Paediatr 1962; 51:161-4. (Abstract) / (PDF)
4-Rowe MI, Clatworthy HW. Incarcerated and strangulated hernias in children. A statistical study of high-risk factors. Arch Surg 1970; 101:136-9. (Abstract) / (PDF)
5-Lau ST, Lee YH, Caty MG. Current management of hernias and hydroceles. Semin in Pediatr Surg 2007; 16:50-7. (Abstract) / (Full Text)/ (PDF)
6-Rowe MI, Clatworthy HW Jr. The other side of the pediatric inguinal hernia. Surg Clin North Am. 1971; 51:1371-6. (Abstract)
7-Rowe MI, Marchildon MB. Inguinal hernia and hydrocele in infants and children. Surg Clin North Am 1981; 61: 1137-45. (Abstract)
8-Snyder W Jr, Greaney E Jr: Inguinal hernia. In: Benson C, Mustard W, Ravitch M, et al (eds): Pediatric surgery. Chicago, Year Book Medical Publishers, 1962; 573-87.
9-Glick PL, Boulanger SC. Inguinal hernias and hydroceles. In: Grosfeld JL, O’Neill JA Jr, Fonkalsrud EW, Coran AG (eds). Pediatric Surgery, 6th ed., Philadelphia: Mosby-Elsevier, 2006, 1172-92.
10-Martin LC, Share JC, Peters C, Atala A. Hydrocele of the spermatic cord: embryology and ultrasonographic appearance. Pediatric Radiol 1996; 26:528-30. (Abstract) / (PDF)
11-Schneck FX, Bellinger MF. Abnormalities of the Testes and Scrotum and their Surgical Management. In: Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (eds). Campbell-Walsh Urology, 9th ed., Philadelphia: Saunders, 2007; 3761-98.
12-Scorer CG, Farrington GH. Congenital deformities of the testis and epididymis. New York, Appleton-Century-Crofts, 1971.
13-Cryptorchidism, 1992. Cryptorchidism: A prospective study of 7500 consecutive male births, 1984-8. John Radcliffe Hospital Cryptorchidism Study Group. Arch Dis Child 1992; 67:892-9. (Abstract) / (Full Text)/ (PDF)
14-Berkowitz GS, Lapinski RH, Dolgin SE, Gazella JG, Bodian CA, Holzman IR. Prevalence and natural history of cryptorchidism. Pediatrics 1993; 92:44-9.
15-Thong M, Lim C, Fatimah H. Undescended tests: Incident in 1.002 consecutive male infants and outcome at 1 year of age. Pediatric Surg Int 1998; 13: 37-41. (Abstract)
16-Berkowitz GS, Lapinski RH. Risk factors for cryptorchidism: A nested case-control study. Paediatr Perinatal Epidemiol 1996; 10:39-51. (Abstract)
17-Wenzler DL, Bloom DA, Park JM. What is the rate of spontaneous testicular descent in infants with cryptorchidism?. J Urol 2004; 171: 849-51. (Abstract) / (Full Text)/ (PDF)
18-Walsh TJ, Dall’Era MA, Croughan MS, Carroll PR, Turek PJ. Prepubertal orchiopexy for cryptorchidism may be associated with lower risk of testicular cancer. J Urol 2007; 178: 1440-6. (Abstract) / (Full Text)/ (PDF)
19-Wyllie GG. The retractile testis. Med J Aust 1984; 140: 403-5. (Abstract)
20-Rusnack SL, Wu HY, Huff DS, et al. The ascending testis and the testis undescended since birth share the same histopathol-ogy. J Urol 2002; 168: 2590-1. (Abstract) / (Full Text)/ (PDF)
21-Ritchey ML, Shamberger RC. Pediatric Urologic Oncology. In: Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (eds). Campbell-Walsh Urology, 9th ed., Philadelphia: Saunders, 2007; 3871-906.
22-Li FP, Fraumeni JF. Testicular cancers in children: epidemiological characteristics. J Natl Cancer Inst 1972; 48: 1575-81. (Abstract)
23-Haas RJ, Schmidt P. Testicular germ-cell tumors in childhood and adolescence. World J Urol 1995; 13: 203-8. (Abstract)
24-Pohl HG, Shukla AR, Metcalf PD, et al. Prepubertal testicular tumors: actual prevalence rate of histological types. J Urol 2004; 172:2370-2. (Abstract) / (Full Text)/ (PDF)
25-Ramani P, Yeung CK, Habeebu SS. Testicular intratubular germ cell neoplasia in children and adolescents with intersex. Am J Surg Pathol 1993; 17: 1124-33. (Abstract)
26-Schellhas HF. Malignant potential of the dysgenetic gonad. II. Obstet Gynecol 1974; 44:455-62. (Abstract)
27-Manuel M, Katayama PK, Jones HW Jr. The age of occurrence of gonadal tumors in intersex patients with a Y chromosome. Am J Obstet Gynecol 1976; 124: 293-300. (Abstract)
28-Aarskog D. Clinical and cytogenetic studies in hypospadias. Acta Paediatr Scand Suppl 1970; 203: 203:1+. (Abstract)
29-Wood HM, Elder JS. Cryptorchidism and testicular cancer: separating fact from fiction. J Urol 2009; 181: 452-61. (Abstract) / (Full Text)/ (PDF)
30-Sugita Y, Clarnette TD, Cooke-Yarborough C, Chow CW, Waters K, Hutson JM. Testicular and paratesticular tumors in children: 30 years’ experience. Aust NZJ Surg 1999; 69: 505-8. (Abstract) / (Full Text)/ (PDF)
31-Oottamasathien S, Thomas JC, Adams MC, DeMarco RT, Brock JW 3rd, Pope JC 4th. Testicular tumors in children: a single-institutional experience. BJU Int 2007; 99:1123-6. (Abstract) / (Full Text)/ (PDF)
32-Çiftçi AO, Bingöl-Koloğlu M, Fienocak ME, Tanyel FC, Büyükpamukçu M, Büyükpamukçu N. Testicular tumors in chill. J Pediatr Surg 2001; 36: 1796-801. (Abstract) / (Full Text) / (PDF)
33-Trailer A, Blanc G, Stark E, Haben B, Treiyer E, Steffens J. Prepubertal testicular tumors: frequently overlooked. J Pediatr Urol 2007; 3:480-3. (Abstract) / (Full Text)/ (PDF)
34-Coley BD. Sonography of pediatric scrotal swelling. Semin Ultrasound CT MR 2007; 28:297-306. (Abstract) / (Full Text)/ (PDF)
35-Huddart SN, Mann JR, Gornall P, et al. The UK Children’s Cancer Study Group: Testicular malignant germ cell tumors 1979-1988. J Pediatr Surg 1990; 25: 406-10. (Abstract)
36-Metcalfe PD, Farivar-Mohseni H, Farhat W, McLorie G, Khoury A, Bägli DJ. Pediatric testicular tumors: contemporary incidence and efficacy of testicular preserving surgery. J Urol 2003; 170:2412-6. (Abstract) / (Full Text)/ (PDF)
37-Shukla AR, Woodard C, Carr MC, et al. Experience with testicular sparing surgery for testicular teratoma. J Urol 2004; 171:161-3. (Abstract) / (Full Text)/ (PDF)
38-Marshall S, Lyon RP, Scott MP. A conservative approach to testicular tumors in children: 12 cases and their management. J Urol 1983; 129: 350-1. (Abstract)
39-Altadonna V, Snyder HM 3rd, Rosenberg HK, Duckett JW. Simple cysts of the testis in children: preoperative diagnosis by ultrasound and excision with testicular preservation. J Urol 1988; 140: 1505-7. (Abstract)
40-Rushton HG, Belman AB, Sesterhenn I, Patterson K, Mostofi FK. Testicular sparing surgery for prepubertal teratoma of the testis: a clinical and pathological study. J Urol 1990; 144: 726-30. (Abstract)
41-Pearse I, Glick RD, Abramson SJ, Gerald WR, Shamberger RC, La Quaglia MP. Testicular-sparing surgery for benign testicular tumors. J Pediatr Surg 1999; 34: 1000-3. (Abstract) / (PDF)
42-Valla JS, for the Group D’Etude en Urologie Pédiatrique. Testicular- sparing surgery for benign testicular tumors in children. J Urol 2001; 165:2280-3. (Abstract) / (Full Text)/ (PDF)
43-Lamm DL, Kaplan GW. Urological manifestations of Burkitt’s lymphoma. J Urol 1974; 112: 402-5. (Abstract)
44-Steeno OP. Varicocele in the adolescent. Adv Exp Med Biol 1991; 286:295-321. (Abstract)
45-Kass EJ. The evaluation and management of the adolescent with a varicocele. AUA Update series 1990; 12:90-5.
46-Rabinowitz R. The importance of the cremasteric reflex in acute scrotal swelling in children. J Urol 1984; 132:89-90. (Abstract)
47-Eaton SH, Cendron MA, Estrada CR, et al. Intermittent testicular torsion: diagnostic features and management outcomes. J Urol 2005; 174: 1532-5. (Abstract) / (Full Text)/ (PDF)
48-Personal Shortliffe LM. Infection and Inflammation of the Pediatric Genitourinary Tract. In: Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (eds). Campbell-Walash Urology. Philadelphia: Saunders, 9th ed., 2007, 3232-68.
49-Freedman A: Urinary tract infection in children. In: Litwin M, Saigal CS (eds). Urologic Diseases of America. US Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. Publication No. 04-5512, Washington, DC: Government Publishing Office; 2004: 213-32.
50-Tarantino L, Giorgio A, de Stefano G, Farella N. Echo color Doppler findings in postpubertal mumps epididymo-orchitis. J Ultrasound Med 2001; 20: 1189-95. (Abstract) / (PDF)
51-Williams CB, Litvak AS, McRoberts JW. Epididymitis infancy. J Urol 1979 ; 121:125-6. (Abstract)
52-Merlini E, Rotundi F, Seymandi PL, Canning DA. Acute epi- didymitis and urinary tract anomalies in children. Scand J Urol Nephrol 1998; 32:273-5. (Abstract) / (PDF)
53-QVIST O. Swelling of the scrotum in infants and children, and non-specific epididymitis; a study of 158 cases. Acta Chir Scand 1956; 110: 417-21. (Abstract)
54-Clark WR, Kramer SA. Henoch-Schönlein purpura and the acute scrotum. J Pediatr Surg 1986; 21: 991-2. (Abstract)